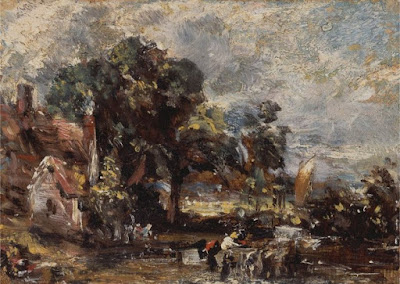The earth pigments are our oldest colors, and they’ve served humanity well.
| Dry Wash, oil on canvasboard, 12X16, $1159 unframed. |
Years ago, I met an artist in Taos who told me that he never used the earths—siennas and umbers—in his paintings. I don’t remember his name, but I vividly remember his rationale. They were too close in color to the rocks of New Mexico. He did better to mix those warm shades.
That is very close to my own rationale for not having greens on my palette. The East is a predominantly-green environment. Using greens straight out of the tube is the best possible way to deaden your painting into a universal dull greenness without variety, sparkle or light.
 |
| Old Barnyard in New Mexico, oil on canvasboard, 12X16, $1159 unframed. |
I’m teaching in New Mexico this week, so I have to adjust my own palette. My reliance on the earths has to ease up. However, my students are all from the south or east, meaning they’ll go back to a green landscape. I want them to take home the logic behind my palette, not an arbitrary rule.
The earth pigments are minerals that have been used in painting since prehistoric times. They’re primarily iron oxides and manganese oxides. We know them as the ochres, siennas and umbers. They’re extremely lightfast. In watercolor they granulate beautifully because of their large particle size. They’re relatively non-toxic* and they’re cheap. Those are all valuable properties to the painter, which is why they’re so widely used.
On the other hand, they look just like the earth because they’re made of dirt and rust (although we synthesize some of them today). They’re complex colors with lots of overtones. In mixtures they stubbornly retain traces of their own character. In a painting predominated by the natural reds and browns of the west, that can get pretty dull, pretty fast. If you want to know how a reliance on the earth pigments will turn out, see Rembrandt—great for Dutch interiors, not so good for American landscape.
 |
| Downdraft snow, oil on canvasboard, 12X16, $1159 unframed. |
The world of greens cannot be simulated by something taken from nature. The pigments that give us green in nature—chlorophylls—are not lightfast. Instead, painters rely on an inorganic compound, chromium oxide green, which we know as viridian or chrome green. Chromium oxide is synthetic, but it does appear in nature as a rare mineral. It is relatively low-stain and inexpensive. It too granulates, which makes it valuable to watercolorists.
Chromium oxide green is a good workhorse pigment, far preferable to the deadening sap green that so many painters love. Sap green started as an unstable extract of buckthorn berries. What we buy today is a convenience mix based on phthalo green. That’s also true of the mixture marketed as ‘viridian hue.’ Paints based on phthalocyanine dyes are very high-stain and have a different color profile than the pigments they’re mimicking. That’s not to disparage the phthalo blue and greens; in themselves they’re lightfast, cheap, and have transformed the modern world.
| Spring thaw along the upper reaches of the Pecos River, oil on canvasboard, 12X16, $1159 unframed. |
Hooker’s Green is another convenience hue. It’s named after an English botanical illustrator, William Hooker, who first put Prussian blue and gamboge together to make a clear, light green. There’s nothing wrong with that mixture—but you should be able to make it yourself, not buy it out of a tube.
That’s true across your palette. You’ll have more flexibility and less expense if you stop buying convenience mixes and ‘hues’.
*Don’t ever fall for the idea that if it’s natural, it’s non-toxic. Mother Nature has hidden a lot of dangerous minerals in this beautiful earth, including cinnabar, galena, lead, asbestos and more.